Body Centred Cubic Cell
Examples of metals with the bcc structure are alpha iron tungsten chromium and beta titanium. The Volume of Body Centered Unit Cell formula is defined as cube of the edge length of the body centered unit cell is calculated using Volume 4 Radius of Constituent Particle sqrt.
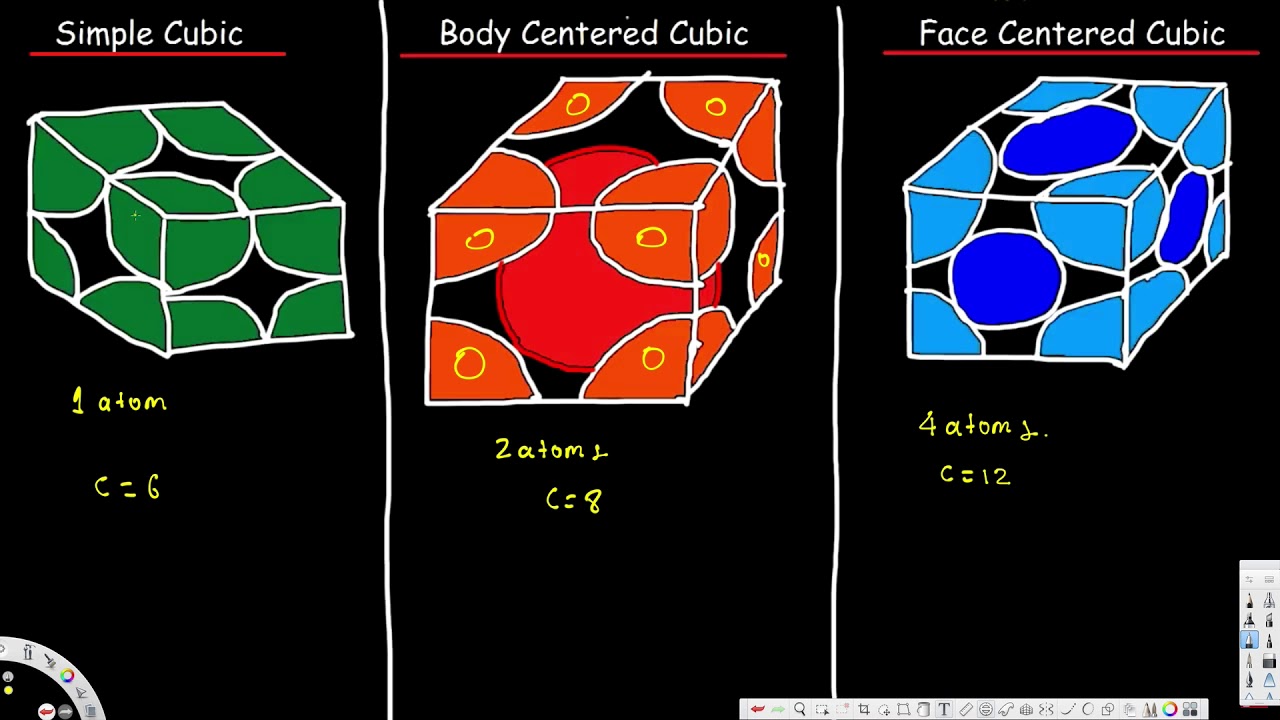
Unit Cell Simple Cubic Body Centered Cubic Face Centered Cubic Cryst Unit Cell Crystal Lattice Structure Nomenclature Chemistry
What is body-centered cubic unit cell.

. What is a body-centered cubic structure. It highlights the key differences between the sim. Body-centered cubic BCC is the name given to.
1 simple cubic 2 face-centered cubic. Some metals crystallize in an arrangement that has a cubic unit cell with atoms at all of the corners and an atom in the center as shown in Figure 2. Eight other unit cells share the atom at the.
Unit cells occur in many different varieties. How do you measure the edge of a body centered cubic. A body-centered cubic unit cell structure consists of atoms arranged in a cube where each corner of the cube shares an atom and with one atom positioned at the center.
This chemistry video tutorial provides a basic introduction into unit cell and crystal lattice structures. Thus the unit cell of each bcc. 4 Atomic packing factor in body centred cubic unit cell is 68 percent.
There is one atom or ion in the center of the unit cell in addition to the corner atoms or ions. Europium is the least abundant lanthanide element with symbol Eu atomic number Z 63 and atomic molar mass 16726 g mol 1. Body-centered cubic BCC is the name given to a type of atom arrangement found in nature.
How do you measure the edge of a body centered cubic. If the atomic radius of vanadium is 134 pm calculate the density of solid vanadium. A 533 gcm3 b 429 gcm3 c 571 gcm3 d 052.
It crystallizes in a body-centered cubic BCC unit cell. There are also atoms or ions. Therefore the total number of atoms in one unit cell is 8 18 1 atom.
Vanadium has a body-centered cubic structure. In the body-centered cubic lattice a total 4 radius one complete atom and two half atoms participate at the body. 5 Relationship between cube edge.
Atomic packing factor in face-centred cubic unit cell has 12. Calculate the crystallographic radius of a tantalum atomWatch the full video athtt. 425 59 votes.
The body-centered cubic bcc lattice Figure 14b can be obtained by adding a second lattice point at the center of each cubic cell of a simple cubic lattice. As one example the cubic crystal system is composed of three different types of unit cells. A body-centered cubic unit cell structure is composed of atoms organized in a cube with one atom in each corner and one atom in the center.
Tantalum forms a body-centered cubic unit cell with a3302 mathrmpm. A body-centered cubic unit cell structure consists of. In each cubic unit cell there are 8 atoms at the corners.
In the body-centered cubic lattice a total 4 radius one complete atom and two half atoms participate at the body diagonal. Body-centred Cubic Unit Cell BCC source.

Unit Cell Body Centered Cubic Crystal Lattice Structures Physical E Crystal Lattice Structure Unit Cell Nomenclature Chemistry

Unit Cell Chemistry Atomic Radius Density Edge Length Calculations Unit Cell The Unit Atom

Planar Packing Fraction Factor For The Body Centred Cubic 111 Plane Fractions Body Packing
11 7 Structure Of Solids Chemistry Libretexts Unit Cell Material Science Physical Chemistry
No comments for "Body Centred Cubic Cell"
Post a Comment